Paracervical Block
The pain of uterine contractions is conducted through nerves passing
close to the cervix. From there, the pain is conducted through multiple
fibers to the sympathetic chain at L2-L5.
Labor pain can be effectively blocked by interrupting the
transmission of pain sensation as it passes through or close to the
cervix. This is called a paracervical block.
Up to 20cc of 1% Lidocaine is
used. 10 cc is injected on each side of the cervix, usually in divided
doses, at 10 o'clock, 8 o'clock, 2 and 4 o'clock (5 cc in each site).
Usually within 5 minutes, the patient becomes completely pain free. The
block will last 60-90 minutes and can be repeated.
The block is effective when lidocaine reaches the broad ligament.
Injections directly into the cervix may block some pain associated with
dilation, it will fail to block the pain of the uterus contracting. In
theory, you should be able to have a single injection site on each side
of the cervix and the block will be effective. In practice, there is
enough anatomic variation from person to person over the precise
location of the broad ligament that experienced physicians typically
will use divided doses to insure that at least some of the drug will get
to where it's supposed to go.
How to Give a Paracervical
Block
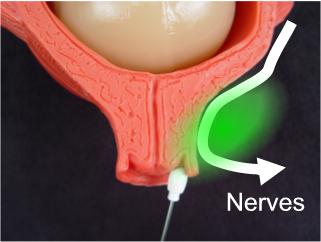
The nerves conducting the pain of labor pass next to the cervix.
Blocking nerve conduction at this point blocks labor pain.
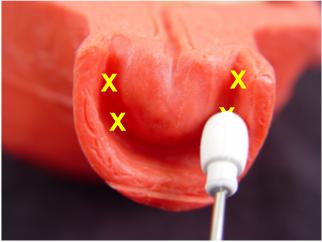
Inject a total of 20 cc of 1% Lidocaine
into the lateral vaginal fornices, with injection sites of 10, 8, 2, and
4 o'clock (5 cc in each site).
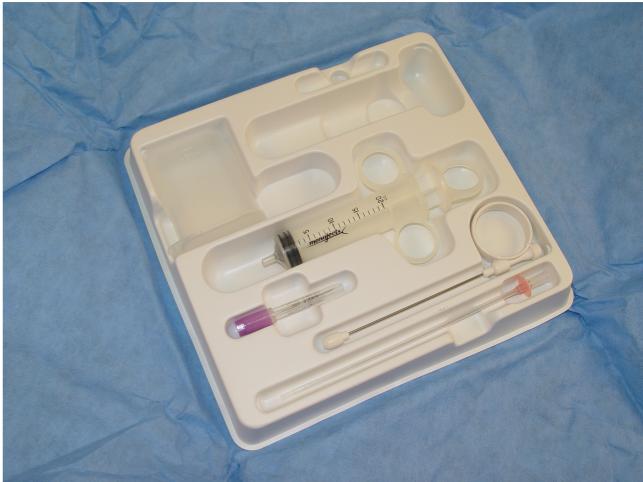
Pre-packaged Pudendal Block and
Paracervical Block Set
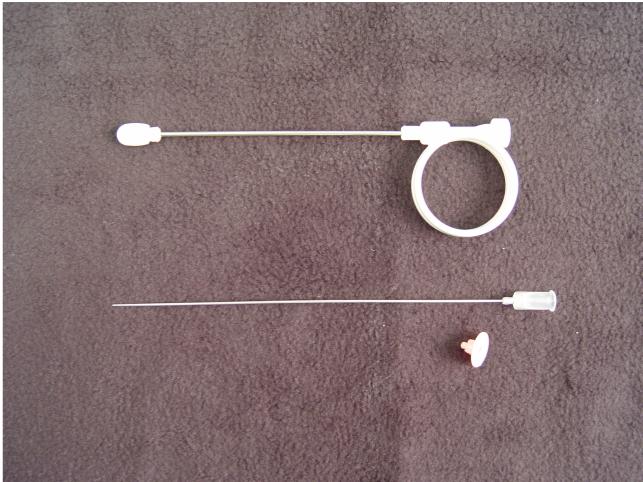
Iowa Trumpet (top), Needle (middle), and
plastic needle spacer to limit depth of penetration of the needle to 5
mm. |
The essential equipment includes and "Iowa Trumpet," a long, hollow,
blunt tube for placing at the injection site, and a matching needle,
slightly longer. Pre-packaged kits typically will include both the Iowa
Trumpet and the needle. Most kits will also have a plastic spacer on the
needle that limits its depth of penetration to 5 mm beyond the Iowa
Trumpet. Removal of the spacer allows for a full 10 mm depth of
penetration (too deep for a paracervical block, but just right for a
pudendal block).
Open the kit and put on sterile gloves. The injection
is done by feel, not by visualization. Use your right hand to examine
the patient, determine the cervical dilatation, and location of the
fetal presenting part. Keeping two fingers of the right hand in the
vaginal, slide the Iowa Trumpet (without the needle) down the length of
your vaginal fingers and direct it into the right lateral vaginal fornix.
It should go in as far as it can go so the tip is resting against the
vaginal mucosa of the later fornix. Adjust the Trumpet so that it is at
the 8 o'clock position.
Once the trumpet is in place, slide the long needle through the Iowa
Trumpet until it is fully seated within the trumpet. At this point, the
tip of the needle will be extending 5 mm beyond the Iowa Trumpet and
just into the paracervical tissues.
Aspirate to make sure you haven't perforated a blood vessel, then
slowly inject 5 cc. Repeat the procedure at the 10 o'clock position.
Move to the opposite side. Switch vaginal examining hands so that the
left hand is in the vagina when injecting on the left side of the
patient. Repeat the procedure, injecting 5 cc at 2 and 4 o'clock.
Complications
The only common complication from this procedure is a post-paracervical
block bradycardia. This usually develops 10 to 20 minutes following
injection, lasts less than 10 minutes, and resolves spontaneously. It is
seen in about 10% (or less) of patients receiving paracervical blocks.
The reason for the bradycardia is probably a direct fetal myocardial
depressive effect of relatively high levels of fetal serum lidocaine.
The base of the broad ligament is very vascular and injection of
lidocaine into this area causes a relatively high, but short-lived surge
in maternal lidocaine levels (below toxic levels). However, because of
the acid-base gradient across the placenta (fetuses are always a little
more "acidy" than their mothers) and the base nature of lidocaine, the
drug is driven across the placenta, concentrating and resulting in even
higher levels of lidocaine in the fetus than in the mother. Depending on
the rate of absorption and the degree of acid-base gradient, the fetal
lidocaine levels can reach toxic levels and have a pharmacologic effect
on the fetal heart. As the lidocaine levels then rapidly fall in the
mother's blood, the fetal levels also fall and the fetal heartbeat
returns to baseline rates. In studies of fetal effects of this
bradycardia, no change in scalp pH was measurable before, during or
after the fetal bradycardia, so long as the bradycardia lasted less than
10 minutes.
Rare complications, such as direct fetal injection and broad ligament
hematoma have been reported.
Patient Selection
Because of the problem of potential fetal bradycardia, the
paracervical block is best reserved for those patients without any
significant abnormality in their fetal monitor tracing. Fetuses who
already have significant variable decelerations or late decelerations
will have a greater than average difference in acidity between the
placenta and the mother and will be expected to be even more likely to
concentrate lidocaine in their bloodstreams. Normal patients, however,
have very little risk of developing a significant bradycardia that
seriously threatens the fetus.
|




